 
For other isotopes, the isotopic mass usually differs and is usually within 0.1 u of the mass number. One unified atomic mass unit is approximately the mass of one nucleon (either a single proton or neutron) and is numerically equivalent to 1 g/mol.įor 12C, the atomic mass is exactly 12u, since the atomic mass unit is defined from it. 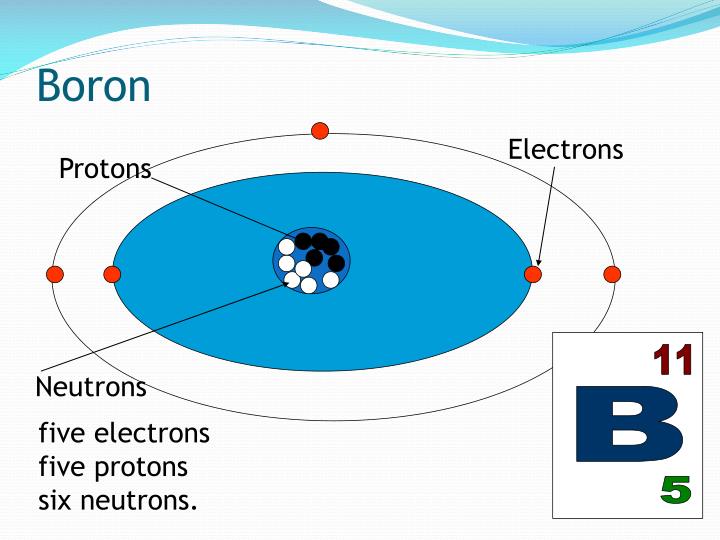
One atomic mass unit is equal to 1.66 x 10 -24 grams.  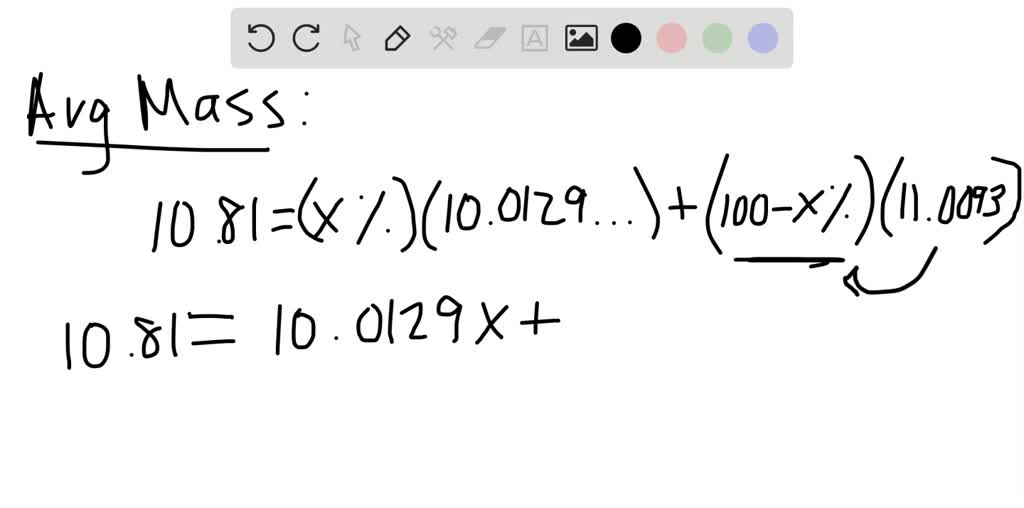
The unit of measure for mass is the atomic mass unit (amu). Therefore this resulting atomic mass is calculated from naturally-occurring isotopes and their abundance. Note that each element may contain more isotopes. How does the atomic number determine the chemical behavior of atoms? Atomic Mass of Boron Since the number of electrons is responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements. 1999‑2023 - All Rights Reserved.Boron is a chemical element with atomic number 5 which means there are 5 protons and 5 electrons in the atomic structure. Retrieved from Ĭopyright © Israel Science and Technology Directory. "Sortable list of elements of the Periodic Table".
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
